Betina Santos Tomaz +
Sarah Gracielly Sena Sousa +
Marcelo Alcantara Holanda +
By the end of this chapter, the reader should be able to:
1. Define capnography and capnometry;
2. Understand the relationship between exhaled CO2, metabolism, ventilation, and pulmonary perfusion;
3. Interpret the normal capnogram and its integration with mechanical ventilation waveforms;
4. Recognize abnormal capnographic patterns and their implications across different clinical scenarios and conditions.
- 1. Capnography: Fundamental Concepts, Operating Principles, and Practical Relevance
- 2. CO2 Physiology and the Relationship Between PaCO2 and ETCO2: Integration of Metabolism, Ventilation, and Pulmonary Perfusion
Metabolism, determines the rate of CO2 production;
Pulmonary perfusion, delivers CO2 to the alveoli;
Alveolar ventilation, removes CO2 from the alveolar space.
- 3. Interpretation of the Normal Capnogram and Its Integration with Mechanical Ventilation Waveforms
Phase I: exhalation of anatomical dead space gas, with no detectable CO2;
Phase II: transition between dead space gas and alveolar gas, characterized by a progressive rise in CO2;
Phase III: alveolar plateau, representing predominantly alveolar gas;
Phase IV: Beginning of inspiration, marked by an abrupt decline in CO2 toward the baseline.
- 4. Capnographic Interpretation Based on Pathophysiological Mechanisms in Common Clinical Conditions
- 5. Summary and Conclusions
- 6. Xlung Trainer Exercises
- 7. References
1. Budania L. Capnography: Principles and application in critical care medicine [Internet]. International Journal of Respiration Care. [cited 2025 Feb 11]. Available from: https://www.ijrc.in/abstractArticleContentBrowse/IJRC/31074/JPJ/fullText
2. Kodali BS. Capnography outside the operating rooms. Anesthesiology. 2014;120(4):955–957.
3. Gravenstein J, Jaffe MB, Paulus DA. Carbon dioxide measurement. In: Gravenstein JS, Jaffe MB, Paulus DA, editors. Capnography. Cambridge, UK: Cambridge University Press; 2011. p. 381–396. doi:10.1017/CBO9780511933837.038.
4. Deranged Physiology. End-tidal capnometry [Internet]. Deranged Physiology. [cited 2025 Feb 11]. Available from: https://derangedphysiology.com/main/cicm-primary-exam/respiratory-system/Chapter-413/end-tidal-capnometry
5. Ortega R, Connor C, Kim S, Djang R, Patel K. Monitoring ventilation with capnography. N Engl J Med. 2012 Nov 8;367(19):e27. doi:10.1056/NEJMvcm1105237.
6. Cook TM, Woodall N, Frerk C. A national survey of the impact of NAP4 on airway management practice in United Kingdom hospitals: closing the safety gap in anaesthesia, intensive care and the emergency department. Br J Anaesth. 2016;117:182-90.
7. West JB. Respiratory Physiology: The Essentials. 10th ed. Philadelphia: Wolters Kluwer; 2016.
8. Nunn JF, Lumb AB. Nunn’s Applied Respiratory Physiology. 8th ed. Philadelphia: Elsevier; 2017.
9. Nassar BS, Schmidt GA. Capnography during critical illness. Chest. 2016;149(2):576-85. 10. Fletcher R. Deadspace, invasive and non-invasive ventilation. Br J Anaesth. 1985;57(3):245-9.
10. Associação de Medicina Intensiva Brasileira (AMIB). Orientações Práticas de Ventilação Mecânica – Interativo SET 11.1. 2024. Available from: https://d1xe7tfg0uwul9.cloudfront.net/amib-portal/wp-content/uploads/2024/09/12100249/Orientacoes-Praticas-de-Ventilacao-Mecanica-Interativo-SET-11-1.pdf (accessed 05 Feb 2026).
11. Kodali BS. Pitfalls of time capnography [Internet]. Capnography.com. 2008 Aug 25 [cited 2026 Feb 10]. Available from: https://www.capnography.com/category/capnopitfalls/capno-pitfalls/
12. Thompson JE, Jaffe MB. Capnographic waveforms in the mechanically ventilated patient. Respir Care. 2005;50(1):100–108.
13. Aminiahidashti H, Shafiee S, Zamani Kiasari A, Sazgar M. Applications of end-tidal carbon dioxide (ETCO2) monitoring in emergency department: a narrative review. Emerg (Tehran). 2018;6(1):e5.
14. Walsh BK, Crotwell DN, Restrepo RD. Capnography/capnometry during mechanical ventilation: 2011. Respir Care. 2011;56(4):503–509. doi:10.4187/respcare.01175.
15. Rasera CC, Gewehr PM. Association between capnogram and respiratory flow rate waveforms during invasive mechanical ventilation. Int J Biosci Biochem Bioinform. 2013;3(2):80–84.
16. Yamanaka MK, Sue DY. Arterial-end-tidal PCO2 difference in respiratory failure. Chest. 1987.
17. Suter PM, Fairley HB, Isenberg MD. Optimum end-expiratory airway pressure in acute pulmonary failure. N Engl J Med. 1975;292:284-289.
18. Amato MBP, Meade MO, Slutsky AS, et al. Driving pressure and survival in ARDS. N Engl J Med. 2015.
19. Sarah Gracielly Sena Sousa. Desenvolvimento de simulador capnométrico à plataforma de simulação Xlung. Início: 2024. Dissertação (Mestrado profissional em Programa de Pós-graduação (PPG) Tecnologia Minimamente Invasiva e Simulação) - Centro Universitário Unichristus.
Capnography is a non-invasive monitoring method that continuously measures the concentration or partial pressure of carbon dioxide (CO2) in both inspired and expired gases throughout the respiratory cycle. The capnograph displays this information as a waveform plotted against time or expired volume. In addition to graphical tracing, capnography provides derived numerical values, the most used being the partial pressure of CO2 at the end of tidal expiration, referred to as ETCO2 (end-tidal carbon dioxide).
Capnometry, in contrast, refers exclusively to the numerical measurement of exhaled CO2 without graphical representation. Thus, while capnometry provides only a single CO2 value, capnography enables dynamic and integrated assessment by reflecting variations in ventilation, pulmonary perfusion, and cellular metabolism through analysis of capnogram morphology. 1, 2 Furthermore, changes in the capnogram waveform may precede alterations in absolute ETCO2 values.2
Infrared absorption spectroscopy is the most widely used technique in capnography. CO2 strongly absorbs infrared light at a wavelength of 4.3 μm. Based on this molecular property, capnographs emit infrared light that is transmitted through a gas sample toward a detector. As the CO2 concentration increases, the intensity of light reaching the detector decreases, enabling measurement of the CO2 fraction in the sample. At this wavelength, interference from other gases commonly present in the breathing mixture (such as water vapor, oxygen, and nitrous oxide), as well as inhaled anesthetic agents, is minimal, allowing reliable application across diverse clinical environments, including operating rooms, emergency departments, and intensive care units.3, 4
Two principal capnograph technologies are available: mainstream and sidestream. Mainstream devices employ sensors positioned directly within the ventilator circuit, enabling real-time CO2 measurement. Sidestream devices continuously aspirate a small gas sample from the respiratory circuit and transport it through a sampling line to an external analyzer, resulting in a short delay in response to changes in CO2 concentration.5 Mainstream devices are particularly suitable for intubated patients receiving mechanical ventilation, ideally integrated with flow, volume, and pressure-time waveforms, as well as volumetric capnography. Sidestream devices, on the other hand, can be coupled to masks or nasal cannulas in non-intubated patients.
Despite the nearly universal use of capnography during anesthesia in high-income countries, its adoption in intensive care units has expanded more recently. In 2011, the Fourth National Audit Project conducted by the Royal College of Anaesthetists and the Difficult Airway Society in the United Kingdom identified an excessive number of airway-related deaths in ICUs, with approximately 80% associated with failure to use capnography or misinterpretation of its data. Publication of these findings led to increased routine capnography use in that setting.6
Currently, capnography is increasingly incorporated into modern mechanical ventilators, becoming part of routine monitoring during invasive ventilatory support. This integration has been associated with improved patient safety, earlier detection of critical airway events, and optimized ventilator settings.2 In this chapter, the focus will be on time-based capnography integrated with invasive mechanical ventilation (MV), excluding volumetric capnography, which warrants dedicated discussion.
CO2 is the primary by-product of aerobic cellular metabolism, being continuously produced in peripheral tissues (V̇CO2) and transported via venous blood to the lungs, where it diffuses into the alveoli and is subsequently eliminated into the atmosphere through alveolar ventilation. In the blood, CO2 is predominantly transported as bicarbonate, with smaller fractions dissolved in plasma and bound to hemoglobin. Pulmonary CO2 elimination depends on the interaction of three fundamental physiological components7:
Alterations in any of these components directly affect CO2 concentrations in arterial blood and exhaled gas.
Arterial CO2 partial pressure (PaCO2), measured by arterial blood gas analysis, serves as the reference standard for evaluating alveolar ventilation. The partial pressure of CO2 at the end of expiration (ETCO2), obtained noninvasively through capnography, reflects, under ideal conditions, alveolar CO2 partial pressure (PACO2).8
When metabolism, pulmonary perfusion, and alveolar ventilation remain relatively stable, PaCO2 and ETCO2 show a strong correlation, with typical reference values of approximately 40 mmHg and 38–35 mmHg, respectively. A small physiological pressure gradient is normally present due to the limited fraction of alveolar dead space, typically 2-5 mmHg, referred to as P(a–et)CO2. Under these conditions, PetCO2 can serve as a continuous, indirect marker of alveolar ventilation, enabling real-time bedside monitoring.7, 9, 10
Dead space refers to the fraction of tidal volume that does not participate effectively in gas exchange and may be classified as anatomical, alveolar, or physiological. An increase in the dead space to tidal volume ratio (VD/VT) reduces CO2 elimination efficiency and leads to widening of the P(a–et)CO2 gradient.9, 10, 11
Figure 1 presents the main indications and benefits of capnography in the invasively mechanically ventilated patient.
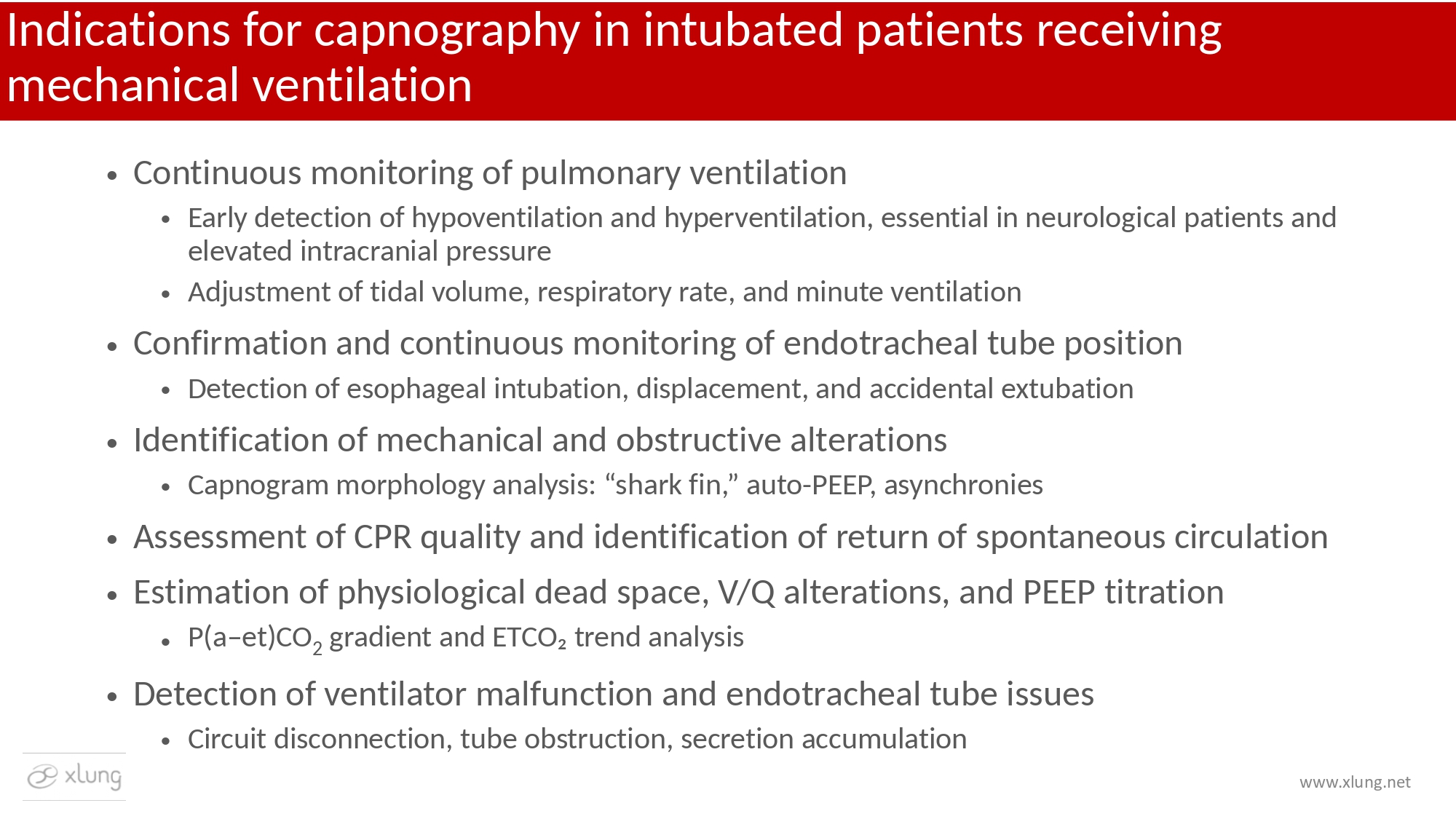
Figure 1. Indications and benefits of capnography integrated with mechanical ventilation waveforms in the intubated patient.
The normal capnogram displays a characteristic waveform composed of four phases:
The end of Phase III corresponds to ETCO2, which under ideal physiological conditions approximates alveolar CO2 partial pressure (PACO2).11-13
Figure 2 illustrates the normal capnogram and its relationship with the physiological and technical determinants involved in its acquisition in the invasively mechanically ventilated patient. Figure 3 presents capnography integrated with the conventional ICU mechanical ventilator waveforms.
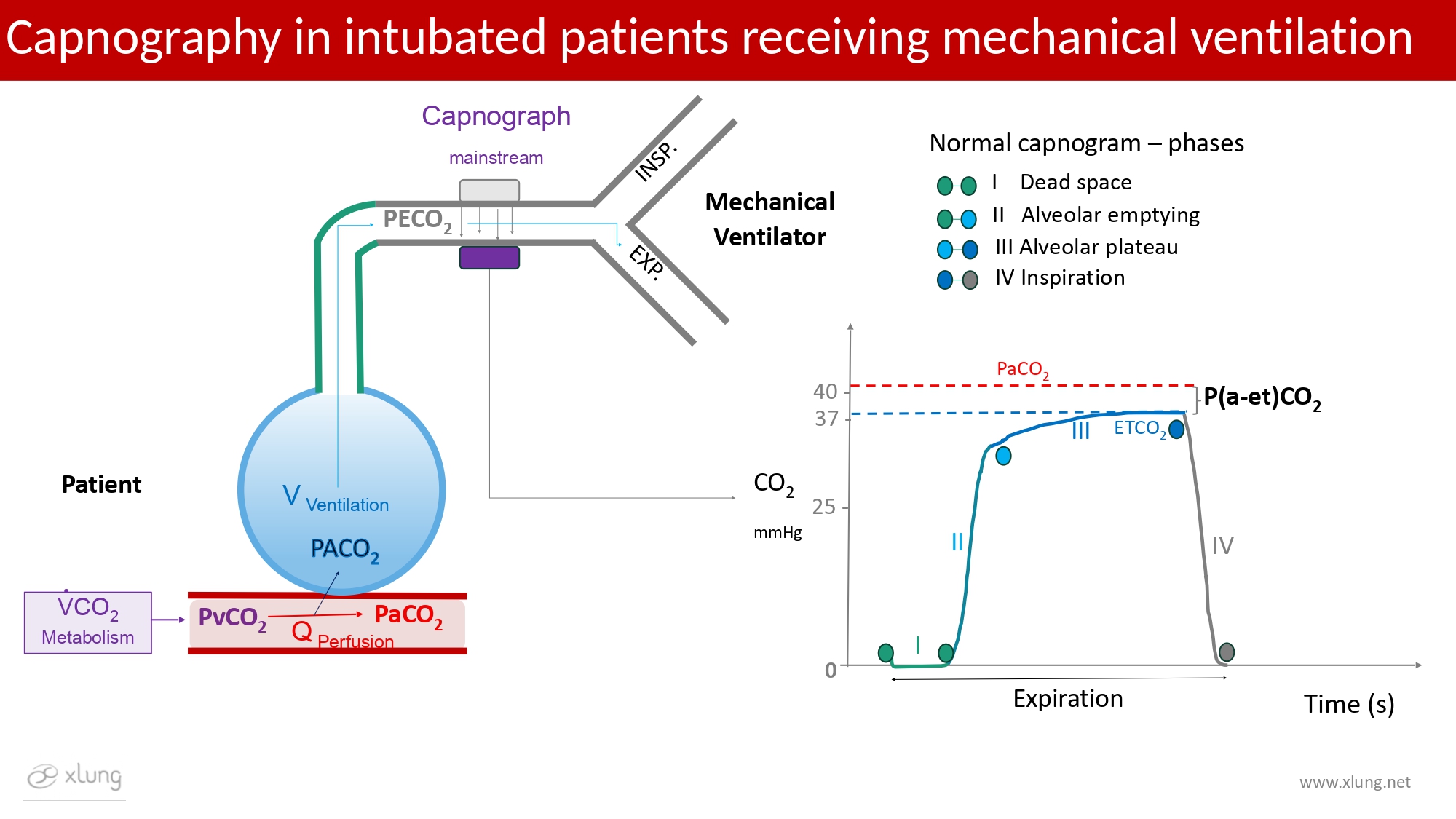
Figure 2. Diagram illustrating mainstream capnography applied to the artificial airway of an intubated patient. Exhaled CO2 partial pressure (PetCO2) results from the interaction between CO2 production (V̇CO2), determined by cellular metabolism, pulmonary perfusion (the hemodynamic component), and alveolar ventilation (the ventilatory component). Continuous measurement of PeCO2 throughout expiration yields a time-based capnogram composed of four phases. Phase I corresponds to exhalation of dead space volume, reflecting the dead space to tidal volume ratio (VD/VT), during which PeCO2 equals zero. Phase II represents the transition as alveolar gas begins to empty, characterized by a rapid rise in PeCO2. Phase III corresponds to the alveolar plateau at end-expiration, where the ETCO2 (or PetCO2) value is obtained. The gradient between arterial and end-tidal CO2 partial pressures, P(a–et)CO2, typically ranges from 2 to 5 mmHg and becomes smaller as ventilation–perfusion (V/Q) matching approaches ideal homogeneity.
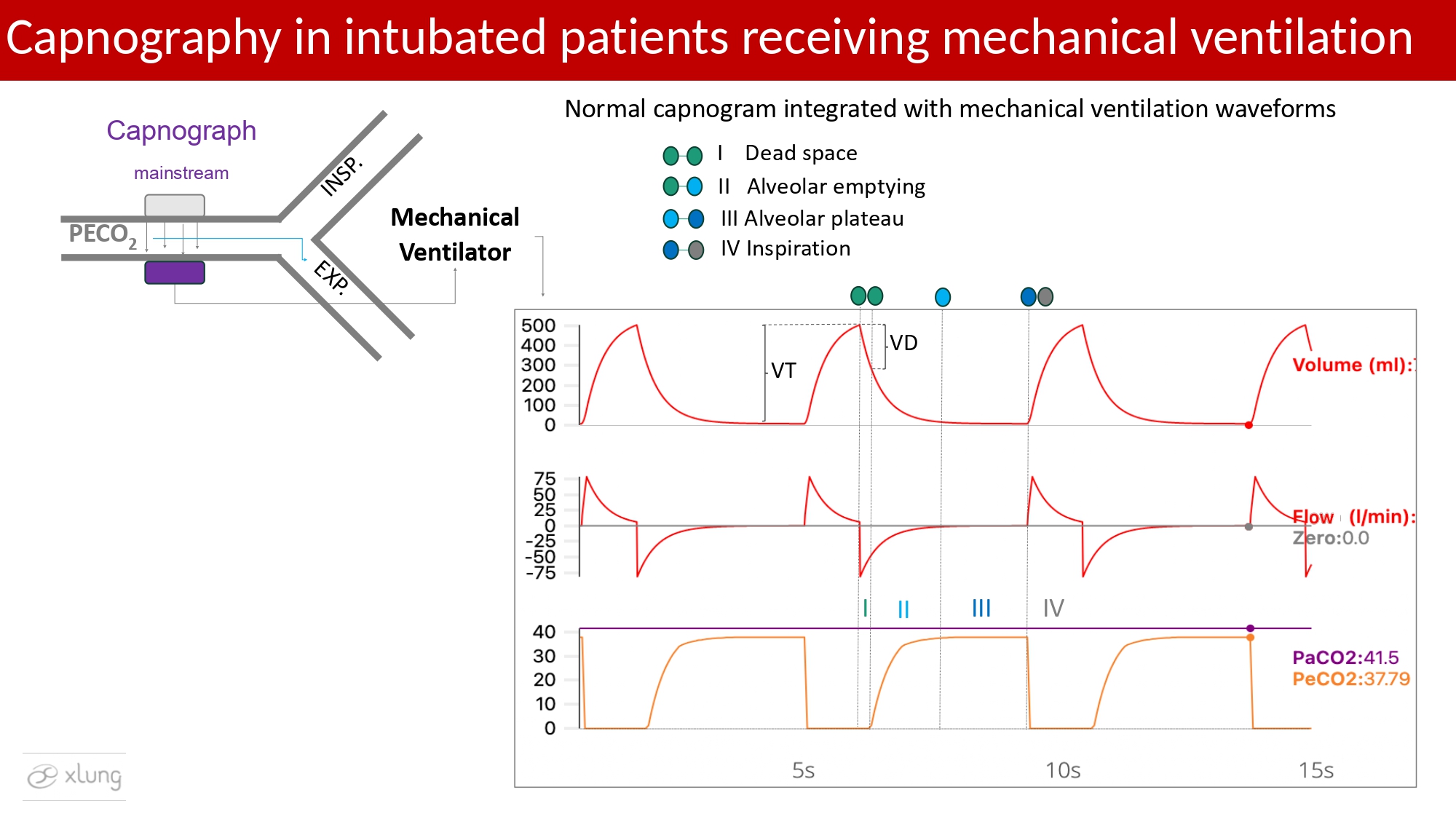
Figure 3. Capnogram waveforms integrated with volume-time and flow-time curves of an ICU mechanical ventilator, demonstrating the four capnographic phases. Phase I corresponds to the exhalation of the dead space volume (VD), when PetCO2 equals zero, and the potential quantification of VD from the tidal volume (VT) curve is possible. Phase II reflects the transition as alveolar gas is exhaled through the proximal airways, with a rapid increase in PetCO2. Phase III represents the alveolar plateau extending to end-expiration, when expiratory flow returns to zero, and PetCO2 is measured immediately before Phase IV, the onset of inspiration. ETCO2 (PetCO2) is shown with a normal physiological gradient of approximately 2–5 mmHg relative to PaCO2, presented here for didactic purposes. Waveforms and data generated using the Xlung simulator.
4.1. Alterations in Metabolic CO2 Production
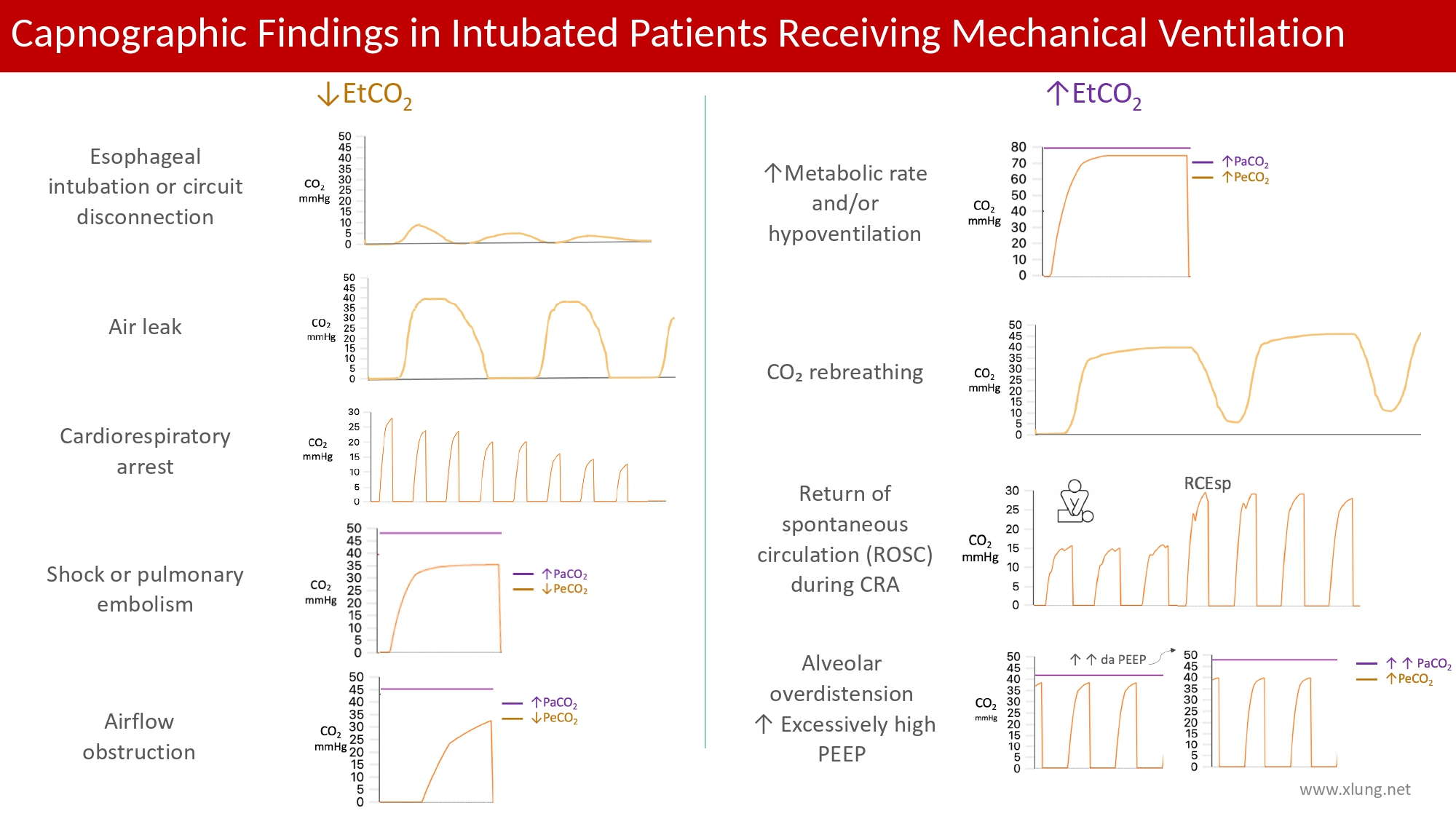
Conditions such as fever, sepsis, seizures, increased muscular activity, and hypercaloric nutritional support elevate metabolic CO2 production, resulting in increased ETCO2 while preserving the P(a–et)CO2 gradient, provided that ventilation and pulmonary perfusion remain stable. The opposite pattern is observed in conditions associated with reduced cellular metabolism, including hypothermia, hypothyroidism, deep sedation, and low caloric intake. Figure 4 illustrates the effects of increased cellular metabolism and its consequences on capnography.14
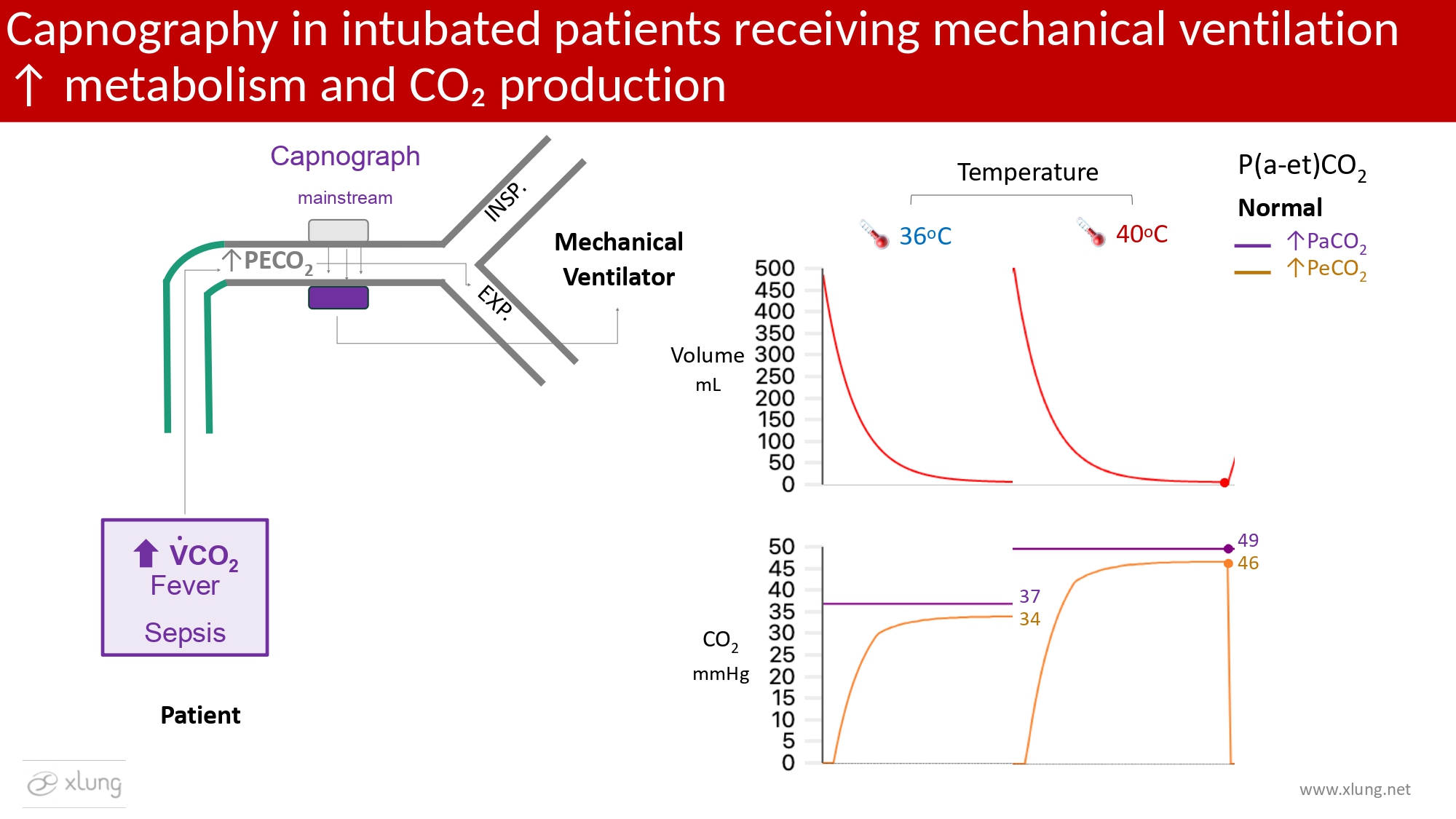
Figure 4. Capnography in intubated patients receiving mechanical ventilation under conditions of increased metabolism and CO2 production.
Fever, sepsis, and other hypermetabolic states increase CO2 production (V̇CO2), resulting in higher mixed expired CO2 values (PECO2). When ventilation–perfusion matching is preserved, both PaCO2 and ETCO2 rise proportionally, maintaining a normal P(a–et)CO2 gradient. For example, an increase in body temperature (36 °C vs. 40 °C) alters capnographic values if the alveolar ventilation does not change. It elevates CO2 levels without changes in the expiratory flow pattern, indicating that alveolar hypoventilation, the primary differential diagnosis, is not responsible for the observed changes. Waveforms and data generated using the Xlung simulator.
4.2. Alveolar Hypoventilation
A reduction in alveolar ventilation, whether due to decreased respiratory rate or reduced tidal volume, leads to CO2 retention, with proportional increases in both PaCO2 and ETCO2. In such cases, the P(a–et)CO2 gradient typically remains within normal limits. Figure 5 presents capnographic findings integrated with the volume-time waveform.14-16
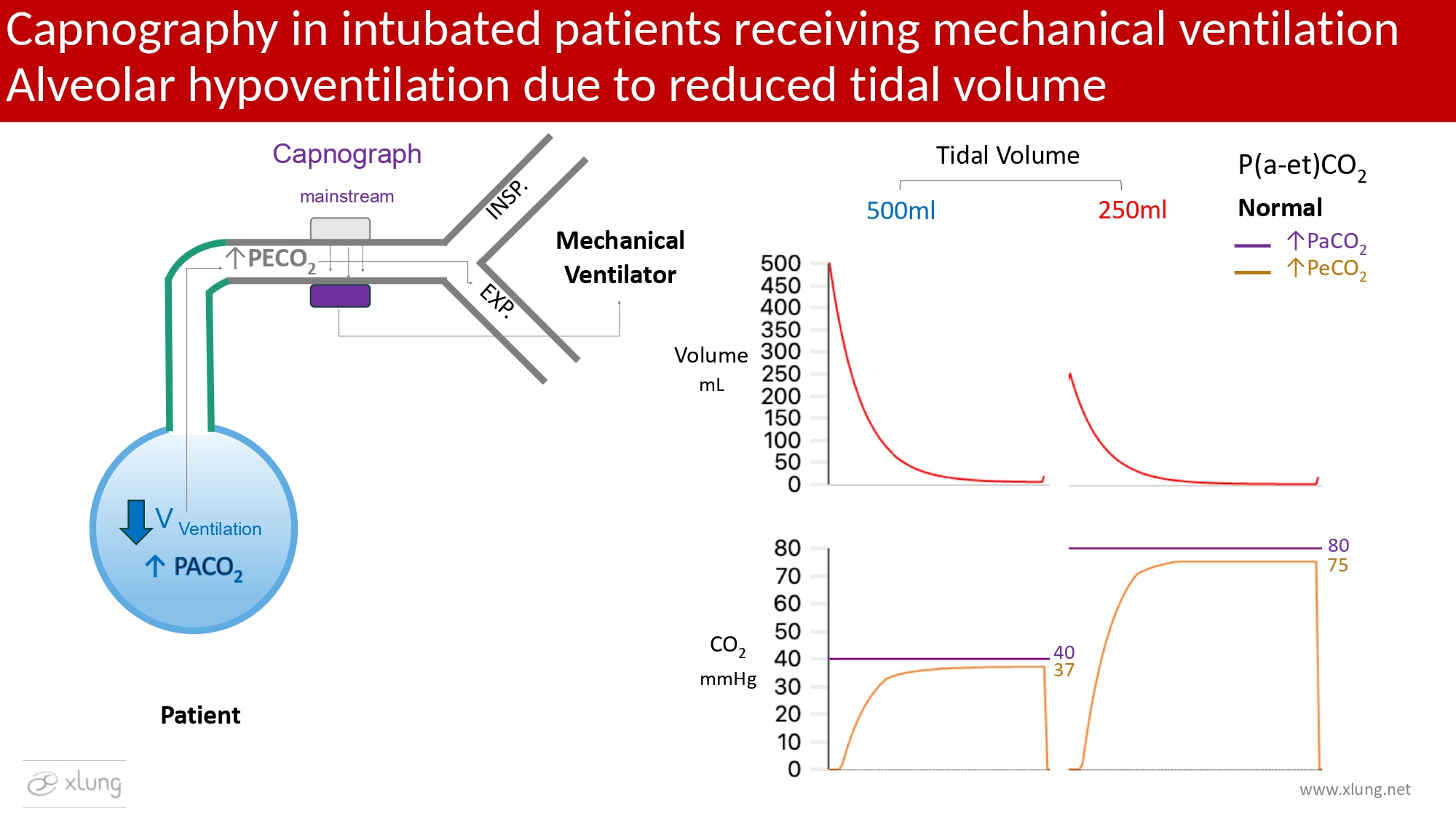
Figure 5. Capnography in intubated patients receiving mechanical ventilation during alveolar hypoventilation secondary to tidal volume reduction. A decrease in tidal volume reduces alveolar ventilation and results in CO2 retention, producing proportional increases in PaCO2 and exhaled CO2 partial pressure (PetCO2). In the presence of preserved ventilation–perfusion matching, the P(a–et)CO2 gradient remains within the physiological range. Integration of physiologically normal-shaped capnograms with volume-time curves demonstrating a reduction in exhaled tidal volume from 500 mL to 250 mL supports hypoventilation as the mechanism responsible for increased PaCO2 and ETCO2. Waveforms and data generated using the Xlung simulator.
4.3. Reduced Pulmonary Perfusion or Circulatory Shock
A reduction in cardiac output or pulmonary perfusion impairs CO2 delivery to the alveoli, leading to a decrease in alveolar CO2 partial pressure (PACO2) and consequently a reduction in ETCO2. This condition is frequently associated with widening of the P(a–et)CO2 gradient and an increase in the VD/VT ratio due to expansion of alveolar dead space.12, 14 Capnographic monitoring may assist in evaluating the response to fluid resuscitation in patients with shock or in evaluating the response to treatment in conditions such as severe pulmonary embolism.
Figure 6 illustrates the effects of reduced pulmonary perfusion on capnography and CO2 values.
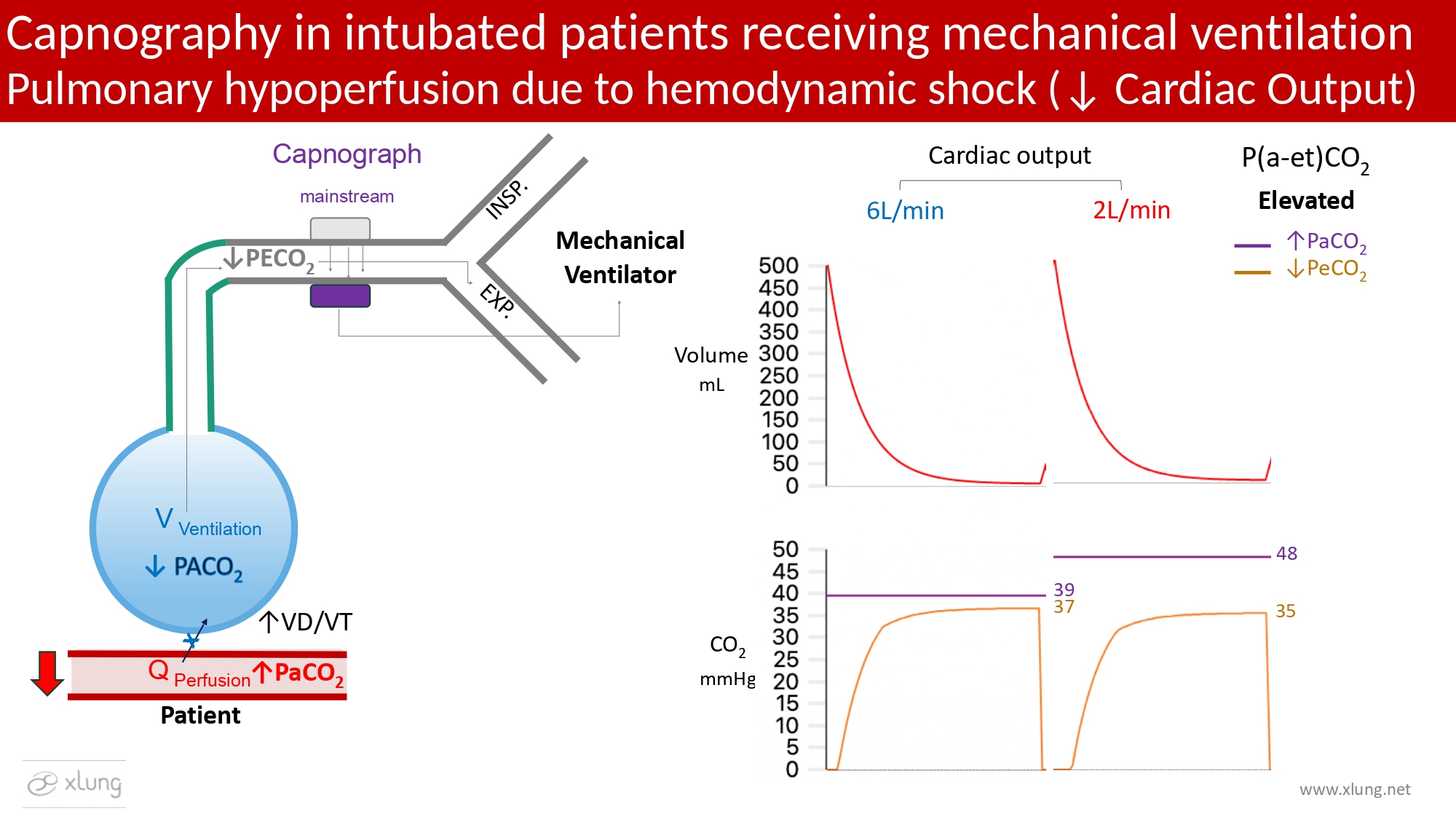
Figure 6. A decrease in cardiac output results in impaired pulmonary perfusion and reduced CO2 delivery to the alveoli, leading to lower exhaled CO2 (ETCO2) despite preserved or elevated arterial CO2 (PaCO2). This condition is associated with increased physiological dead space (VD/VT) and widening of the P(a–et)CO2 gradient. Capnograms obtained under conditions of normal (6 L/min) and reduced (2 L/min) cardiac output demonstrate a marked reduction in ETCO2 with unchanged mechanical ventilation, highlighting the predominantly hemodynamic contribution to the observed capnographic alterations. Waveforms and data generated using the Xlung simulator.
4.4. Airflow Limitation and Auto-PEEP
Obstructive airway diseases, characterized by increased airway resistance, promote expiratory flow limitation, air trapping, and the development of auto-PEEP. The capnogram typically exhibits a progressive upward slope in Phases II and, most prominently, Phase III, producing the classic “shark-fin” appearance. This pattern is frequently associated with reduced ETCO2 and increased physiological dead space, as well as widening of the P(a–et)CO2 gradient. This phenomenon is further exacerbated when expiratory time is insufficient, particularly in patients with severe COPD during acute exacerbations, due to marked ventilatory heterogeneity, excessive airway resistance, reduced elastic recoil in the presence of emphysema, and a substantial increase in the expiratory time constant.2, 12
In this context, PetCO2 does not reliably reflect PaCO2 and therefore cannot replace arterial blood gas analysis for accurate evaluation of alveolar ventilation. Conversely, optimization of ventilator settings and/or improvement of the underlying pulmonary condition, resulting in reduced air trapping, decreased auto-PEEP, and in more complete lung emptying during expiration, generally leads to a reduction in P(a–et)CO2 toward the normal range. The opposite pattern is observed with clinical deterioration.
Figure 7 illustrates the effects of increased airway resistance, pulmonary hyperinflation, and auto-PEEP on capnogram morphology and CO2 measurements.
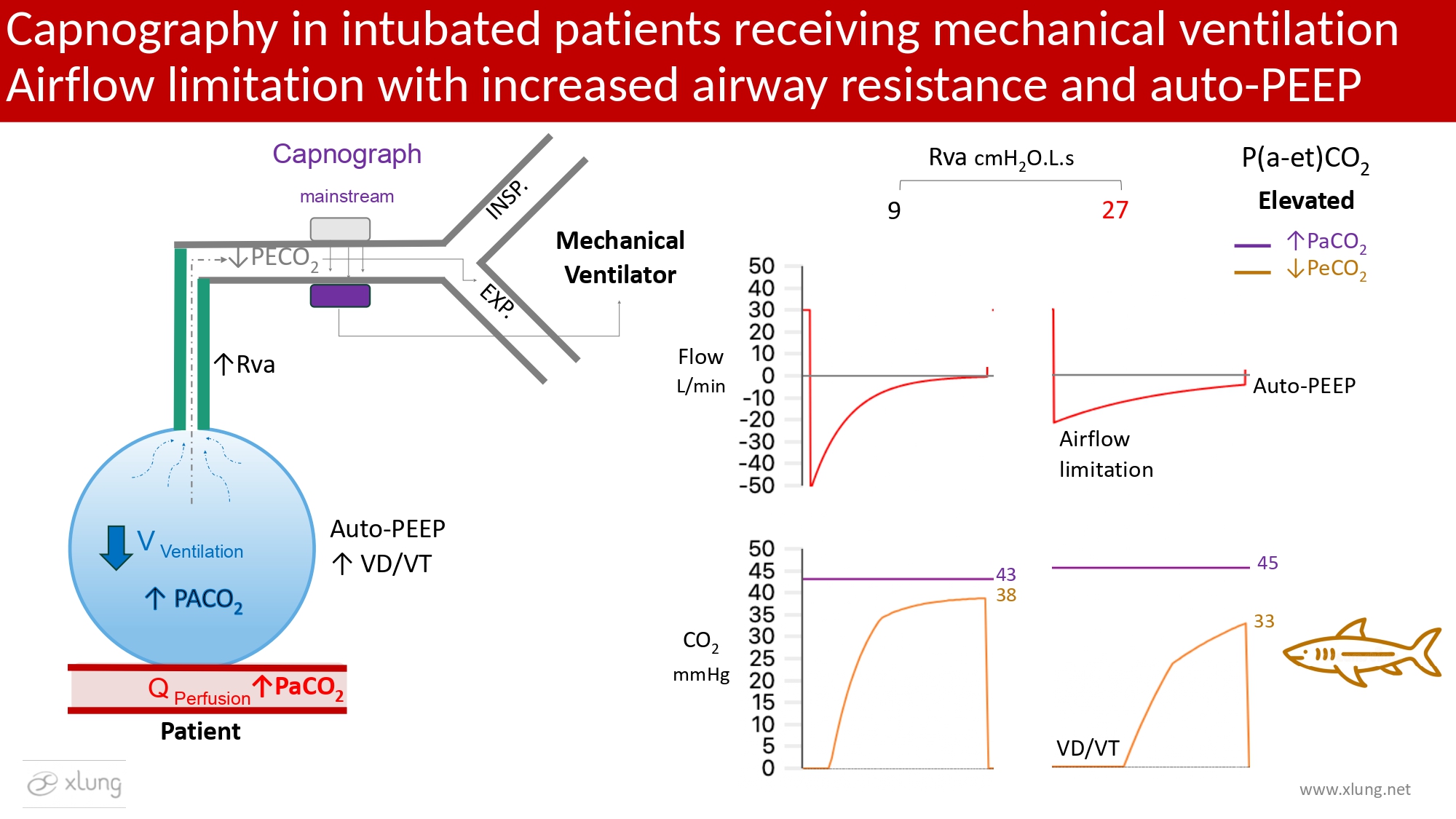
Figure 7. Capnography in an intubated patient receiving mechanical ventilation with airflow limitation, increased airway resistance, and the development of auto-PEEP. Increased airway resistance (Raw) promotes expiratory flow limitation, impairing complete lung emptying. This condition results in dynamic air trapping and the generation of auto-PEEP, evidenced by the inability of expiratory flow to return to zero before the onset of the next inspiratory cycle. Consequently, effective alveolar ventilation is reduced, leading to an increase in the dead space-to-tidal volume ratio (VD/VT). Capnography typically demonstrates reduced ETCO2 values despite a concomitant rise in PaCO2, reflecting alveolar hypoventilation associated with widening of the P(a–et)CO2 gradient. The capnogram exhibits a progressive upward slope of Phases II and III, characteristic of the classic shark-fin pattern seen in airflow obstruction. At the same time, the flow-time waveform confirms the presence of auto-PEEP. Waveforms and data generated using the Xlung simulator.
4.5. Recovery of Respiratory Effort and Patient–Ventilator Asynchrony
During recovery of spontaneous respiratory effort following neuromuscular blockade, ineffective inspiratory efforts that fail to trigger the ventilator may occur. These events can be detected on ventilator waveforms, particularly the flow-time curve, as well as on the capnogram, where they appear as characteristic “curare clefts.” Such events may alter CO2 partial pressure throughout expiration and transiently affect ETCO2 values, potentially producing intermittent variations in the measured ETCO2 and the P(a–et)CO2 gradient. Figure 8 illustrates the capnographic effects of ineffective inspiratory efforts.
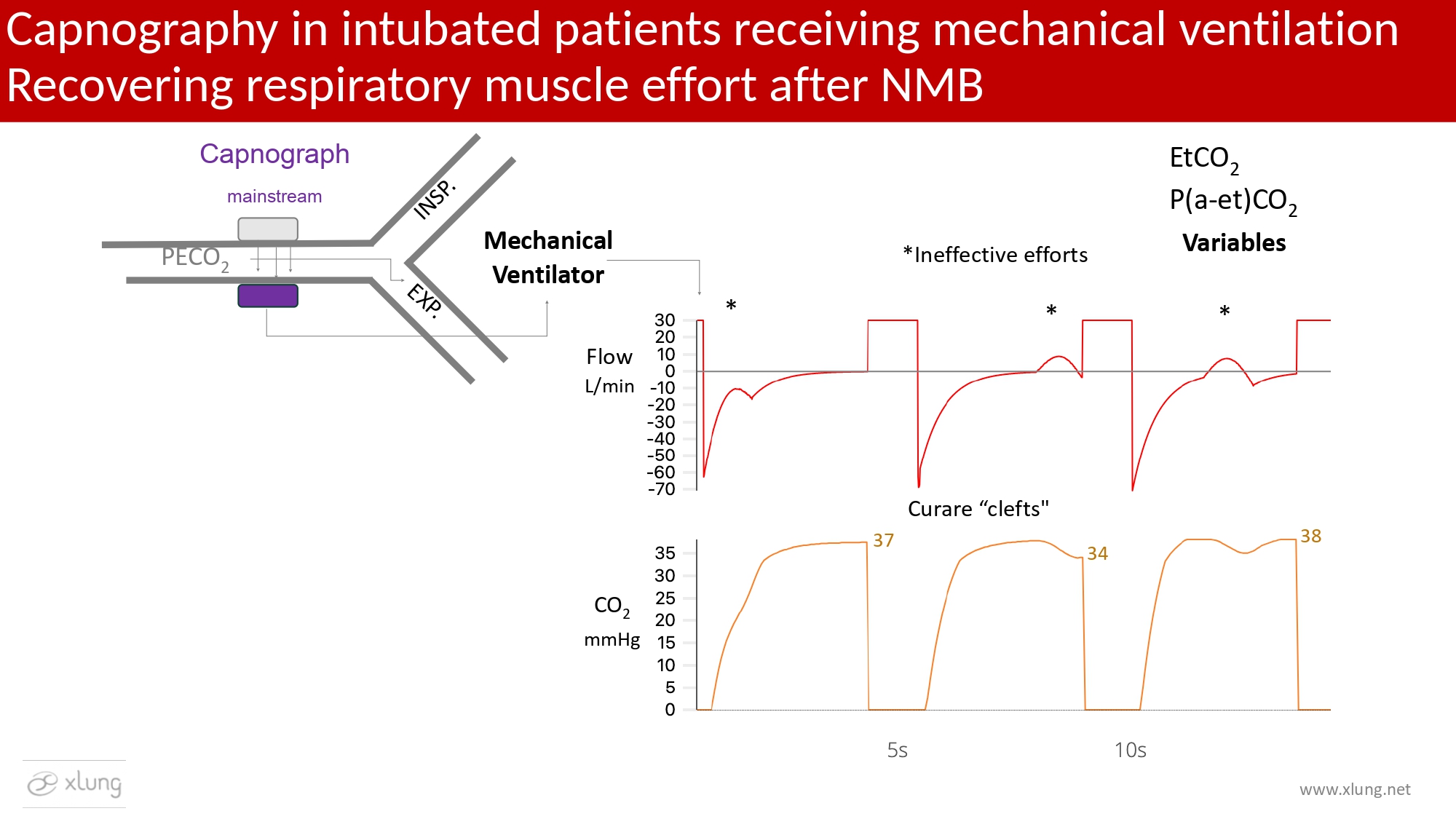
Figure 8. Diagram illustrating ineffective inspiratory efforts, visible on the flow waveform, and their corresponding effects on the capnogram. In this example, waveforms were generated by simulating reduced inspiratory muscle effort, as may occur during partial recovery from neuromuscular blockade (curare cleft). These events may occur near end-expiration, producing transient entrainment of CO2-free gas, thereby altering PetCO2 and potentially affecting ETCO2 measurements. Consequently, variations in the P(a–et)CO2 gradient may also be observed. NMB: neuromuscular blockade. Waveforms and data generated using the Xlung simulator.
4.6. Progression to Cardiorespiratory Arrest
During progression to cardiorespiratory arrest (CRA), a rapid and progressive decline in ETCO2 toward values near zero is observed, despite preservation of the ventilatory pattern. This finding reflects the abrupt interruption of pulmonary perfusion. Within the appropriate clinical context, this pattern should be considered strongly suggestive of CRA. The abrupt reduction in ETCO2 reflects the cessation of CO2 transport from tissues to the lungs and represents an early, highly sensitive marker of circulatory collapse. The capnogram demonstrates progressive loss of Phase III and collapse of the alveolar plateau, ultimately resulting in a nearly flat or absent tracing, while the ventilator continues to cycle, indicating absence of effective circulation.12, 15, 16
Figure 9 illustrates the characteristic capnographic findings associated with this critical condition.
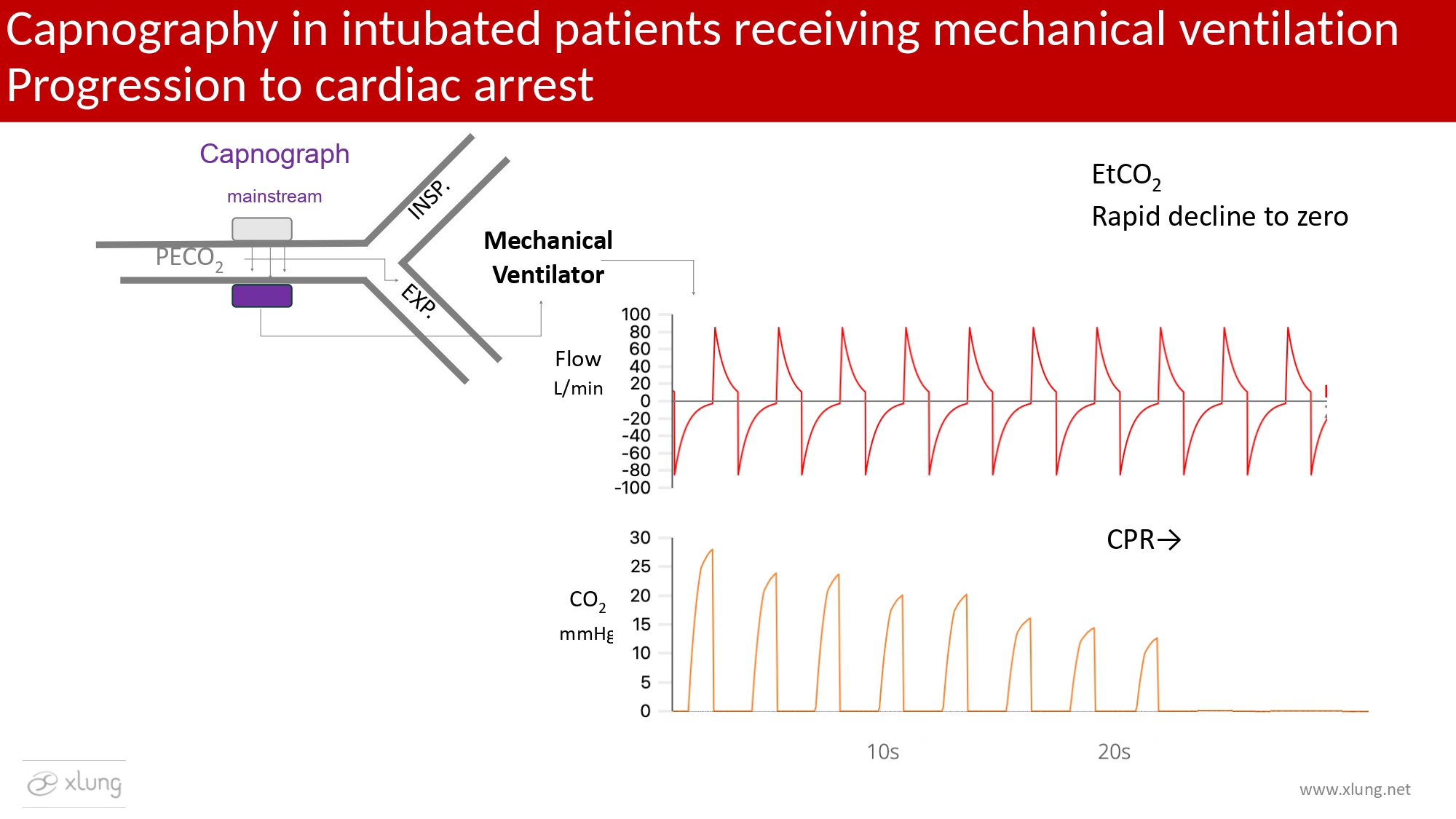
Figure 9. Capnography in an intubated patient receiving mechanical ventilation during progression to cardiorespiratory arrest (CRA). As severe and rapidly progressive hemodynamic deterioration occurs, with abrupt reduction in cardiac output, pulmonary perfusion collapses, leading to a marked reduction in CO2 delivery to the alveoli. Consequently, ETCO2 rapidly declines toward near-zero values despite preservation of the ventilatory pattern, as evidenced by the maintained flow-time waveform.
4.7. Capnography During Cardiopulmonary Resuscitation and Return of Spontaneous Circulation
During cardiopulmonary resuscitation (CPR), ETCO2 directly reflects pulmonary blood flow generated by chest compressions and serves as an indirect marker of chest compression quality. Return of spontaneous circulation (ROSC) is characterized by a sudden and sustained increase in ETCO2, representing recovery of cardiac output and pulmonary perfusion. This parameter is particularly valuable for guiding and monitoring resuscitation efforts. Effective chest compressions are typically associated with ETCO2 values between 10 and 20 mmHg, with higher values within this range generally indicating better perfusion.12, 14 Capnography is increasingly recognized as an essential tool for assessing CPR effectiveness and early identification of ROSC. Figure 10 presents capnographic waveforms before, during varying compression effectiveness, and following ROSC.
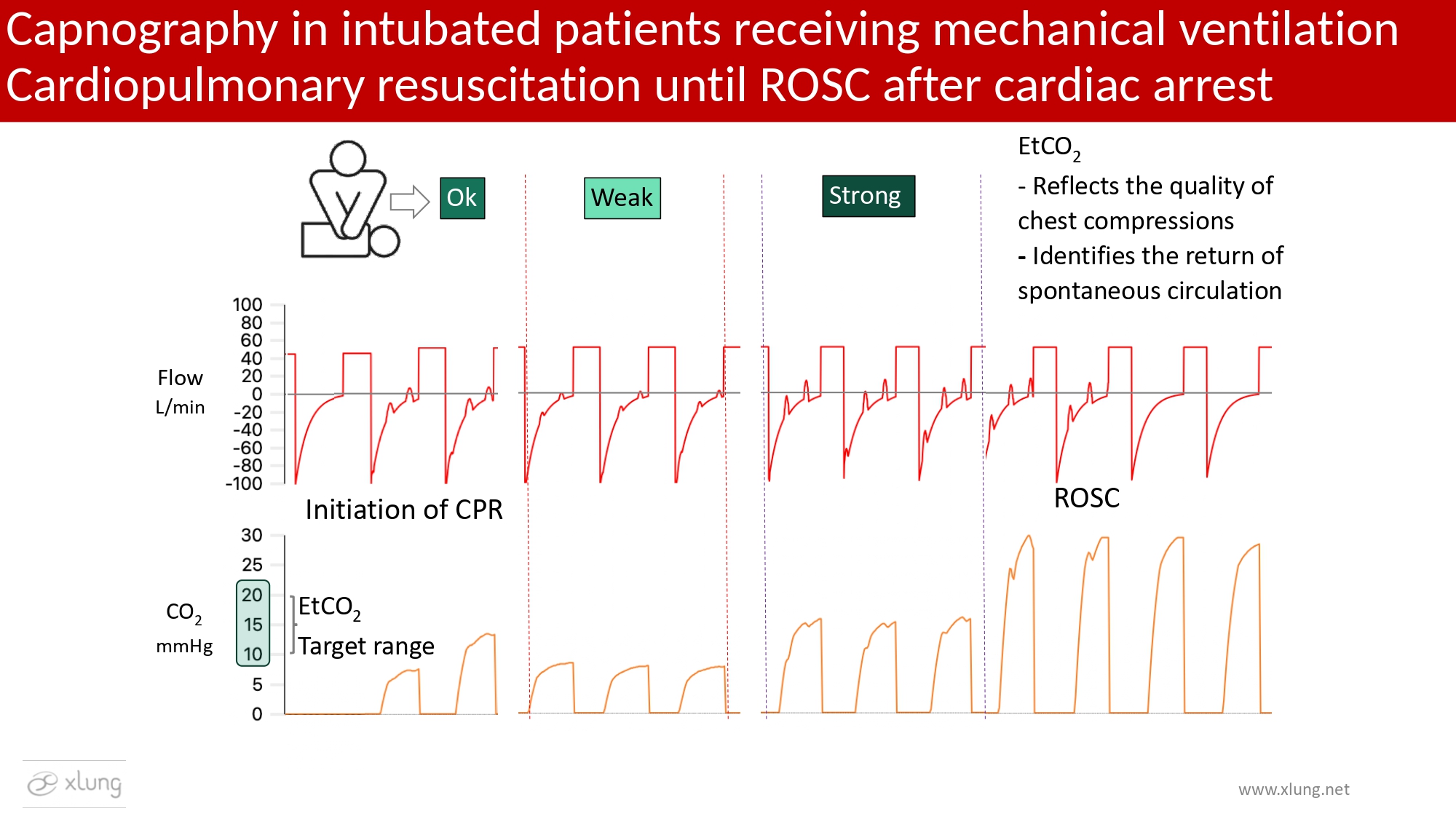
Figure 10. Capnography in an intubated patient receiving mechanical ventilation during cardiopulmonary resuscitation, from cardiorespiratory arrest (CRA) through initiation of chest compressions to return of spontaneous circulation (ROSC). During CRA, ETCO2 values directly reflect pulmonary blood flow generated by chest compressions, functioning as an indirect marker of chest compressions quality. Ineffective compressions are associated with low ETCO2 values, whereas more effective compressions produce progressive increases in ETCO2 within the target range. A sustained and abrupt rise in ETCO2 represents the classic indicator of ROSC. Flow-time waveforms demonstrate maintenance of ventilatory pattern throughout resuscitation, with oscillations related to chest compressions. At the same time, the capnogram shows the transition from low exhaled CO2 levels during CPR to higher, more stable values after ROSC. Waveforms and data generated using the Xlung simulator.
4.8. Capnography During PEEP Titration in Patients with Acute Respiratory Distress Syndrome (ARDS)
Pioneering studies have evaluated the effects of PEEP titration in ARDS on physiological parameters, including cardiac output and respiratory mechanics. The optimal PEEP for a given patient with ARDS should not be defined solely by improvements in oxygenation, but rather by the level that achieves the best balance between alveolar recruitment and hemodynamic stability, ensuring adequate oxygen delivery to tissues and organs without causing alveolar overdistension. Excessively high PEEP levels tend to increase alveolar dead space due to parenchymal overdistension and/or reduced capillary perfusion. In contrast, insufficient PEEP levels favor alveolar collapse, recurrent expiratory atelectasis, and increased pulmonary shunt. PEEP levels associated with global improvement in respiratory system compliance may approximate the best compromise, minimizing overdistension in normally aerated or hyperaerated lung regions while maintaining alveolar patency in dependent lung zones. This strategy contributes to reducing the risk of ventilator-induced lung injury (VILI – Ventilator-Induced Lung Injury).17-19
Figure 11 illustrates the effects of PEEP titration on driving pressure, a variable reflecting the relationship between tidal volume and respiratory system compliance, and on the P(a–et)CO2 gradient, which reflects alveolar dead space. Both variables may provide complementary physiological information during bedside optimization of mechanical ventilation in ARDS, contributing to a lung-protective strategy.

Figure 11. Capnography in an intubated patient with Acute Respiratory Distress Syndrome (ARDS) receiving mechanical ventilation in volume-controlled ventilation (VCV) mode with tidal volume set at 6 mL/kg predicted body weight during decremental PEEP titration (15 → 10 → 5 cmH₂O). A PEEP level of 10 cmH₂O resulted in the lowest driving pressure and the smallest P(a–et)CO2 gradient, findings consistent with reduced pulmonary overdistension and greater alveolar aeration and improved ventilation–perfusion (V/Q) matching compared with the other tested PEEP levels. Waveforms and data generated using the Xlung simulator.
4.9. Additional Clinical Conditions Commonly Identifiable by Capnography
Capnography enables rapid identification of various clinically relevant conditions and technical problems in intubated patients receiving mechanical ventilation. Abrupt and marked changes in ETCO2 values, baseline shifts, or capnogram morphology can function as early warning signs of potentially serious events, often preceding overt clinical manifestations.
Integrated analysis of the capnogram alongside mechanical ventilator waveforms allows detection of endotracheal tube malposition, circuit disconnections, artificial airway migration, leaks, and malfunction of inspiratory or expiratory valve mechanisms within the ventilator circuit.12 Figures 12 and 13 illustrate representative examples of ventilatory delivery abnormalities, circuit-related disturbances, and airway positioning errors. Such scenarios represent high-risk adverse events that can lead to catastrophic outcomes if not promptly recognized and corrected.
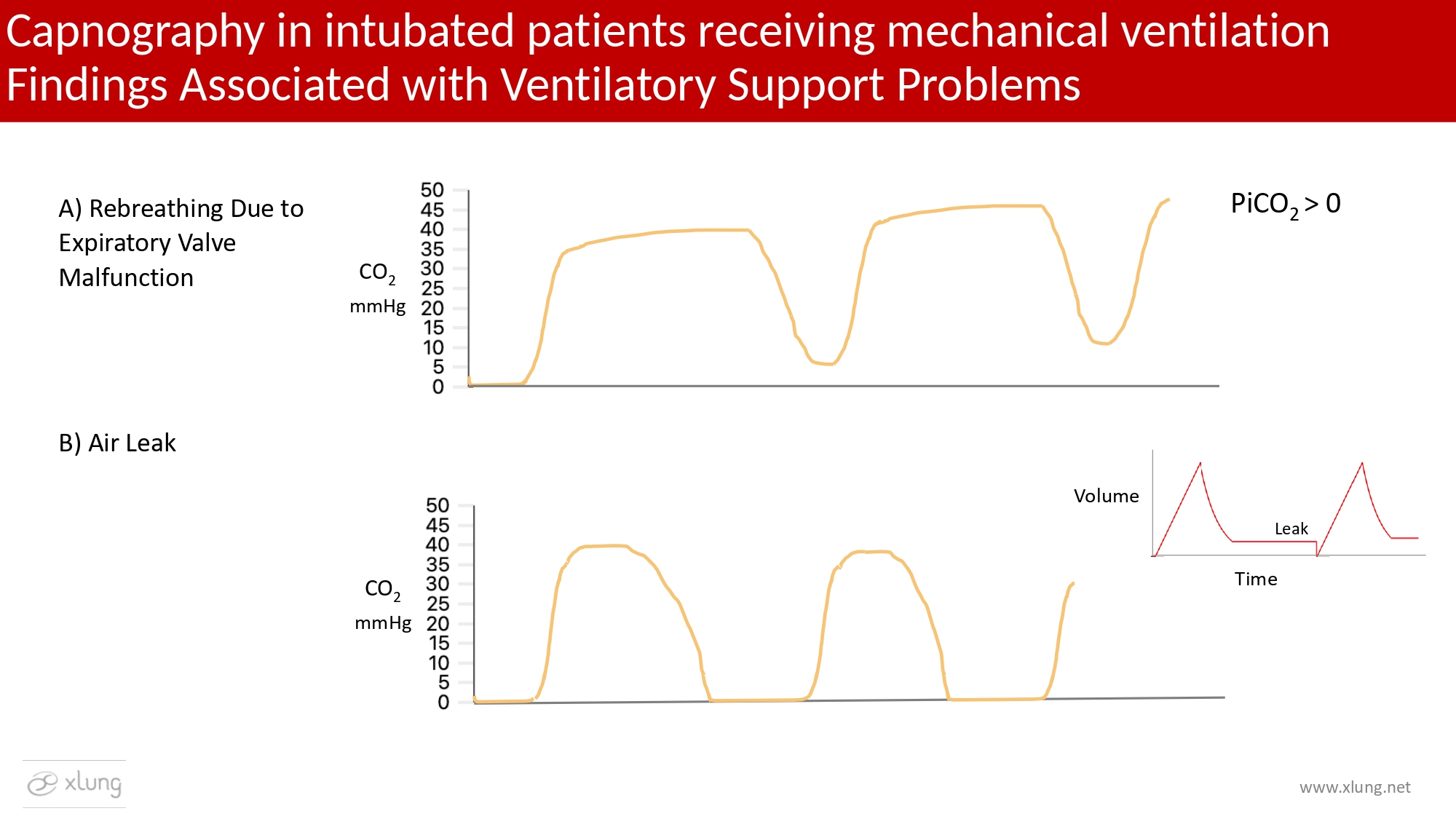
Figure 12. Capnography in intubated patients receiving mechanical ventilation with abnormalities in the control of inspiratory and expiratory gas flow within the ventilator circuit. In A) CO2 rebreathing caused by expiratory valve malfunction results in partial return of exhaled gas at the onset of inspiration (Phases IV and I of the capnogram). An increase in inspired CO2 partial pressure is observed (PiCO2 > 0), proportional to the severity of rebreathing. In the absence of compensatory hyperventilation, PetCO2 may also increase. In B) the presence of a circuit leak causes early reduction of PetCO2, preventing formation of the alveolar plateau and generating a characteristic capnogram morphology.
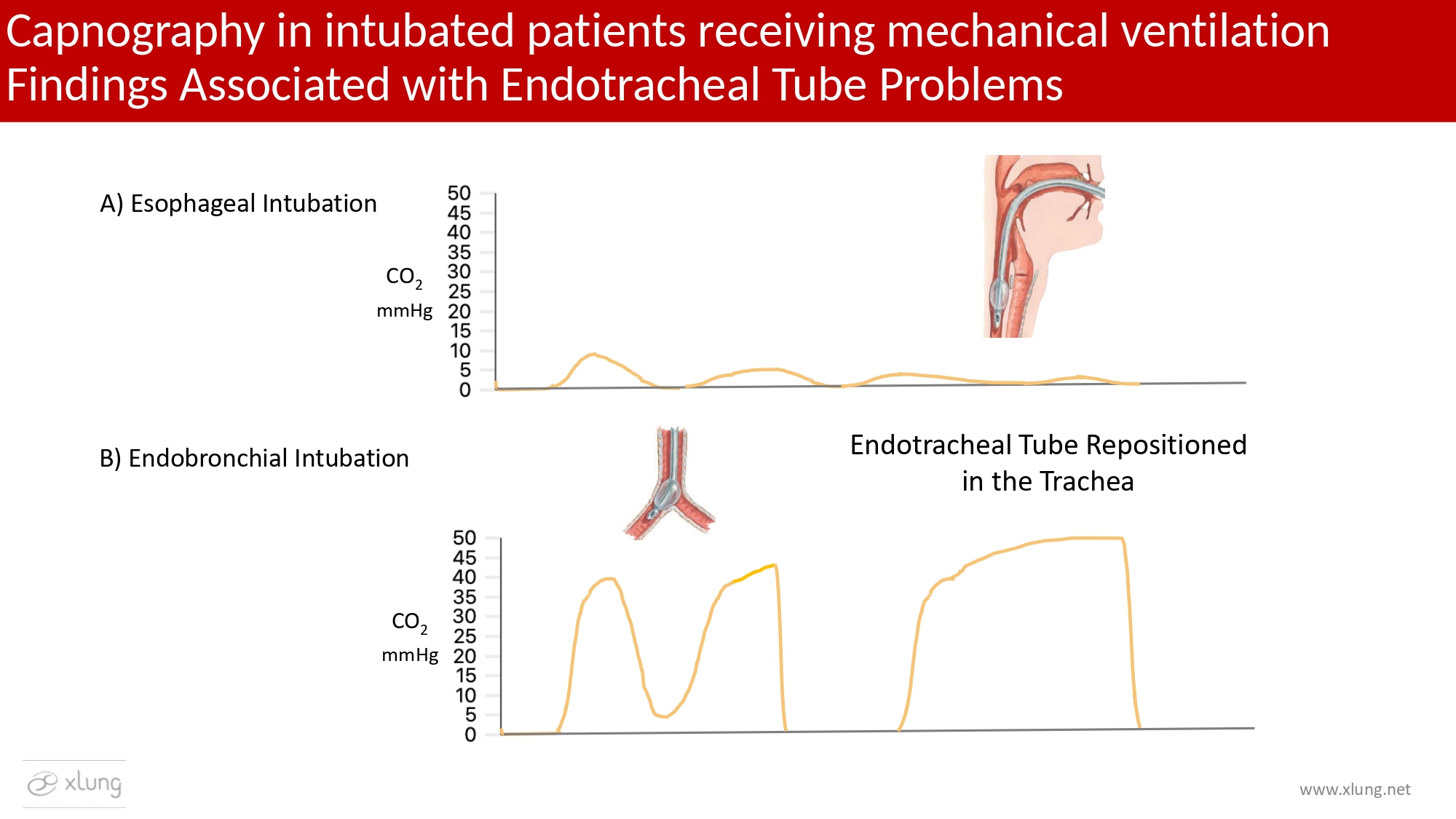
Figure 13. In A) esophageal intubation is characterized by extremely low or absent PetCO2 values, sometimes displaying small transient waveforms due to residual CO2 within the upper gastrointestinal tract immediately after intubation, followed by a flat tracing. The expression “no trace, wrong place” has been proposed to emphasize the urgency of immediate corrective action. In B) migration of the distal end of the endotracheal tube into the right main bronchus produces asymmetric lung emptying, resulting in a biphasic capnogram morphology. This pattern is typically corrected following appropriate repositioning of the distal extremity of the tube within the trachea.
Figure 14 presents an illustrated summary of the main clinical conditions and corresponding capnographic findings discussed throughout this chapter.
Capnography has become firmly established as an essential monitoring modality in intubated patients receiving mechanical ventilation, with expanding applications across diverse clinical scenarios. Scientific evidence supports its value in integrated assessment of ventilation and perfusion, while also facilitating early detection of technical problems with the artificial airway, which may represent high-risk, potentially fatal events if not promptly recognized and corrected. Awareness of its physiological and technical limitations, along with appropriate clinician training, is fundamental for its safe and effective use.
There is growing recognition that capnography education remains insufficient in medical training and across other health professions. In this context, incorporating capnography into the virtual simulators on the Xlung platform is a valuable educational resource that supports the teaching of applied respiratory physiology and mechanical ventilation.20
Ongoing technological advances are expected to expand the integration of capnography into mechanical ventilation systems, making this monitoring tool increasingly accessible, reliable, and clinically relevant.
Access simulation exercises involving capnography application in intubated patients: